We are thrilled to announce the publication of our latest paper in Applied Sciences! Congratulations to Najwa and the team for their work on “The Effect of Boron Oxide on the Structures and Thermal Properties of Phosphosilicate Bioactive Glasses for Metallic Implants’ Coatings.” This research provides valuable insights into enhancing bioactive glass formulations, contributing to the development of advanced coatings for metallic implants. Great job, everyone!
Title: The Effect of Boron Oxide on the Structures and Thermal Properties of Phosphosilicate Bioactive Glasses for Metallic Implants’ Coatings
Authors: Oliver, J.N.; Xie, W; Du, J; Ecker, M.
Journal: Appl. Sci. 2025, 15(3), 1293
DOI: https://doi.org/10.3390/app15031293
Abstract:
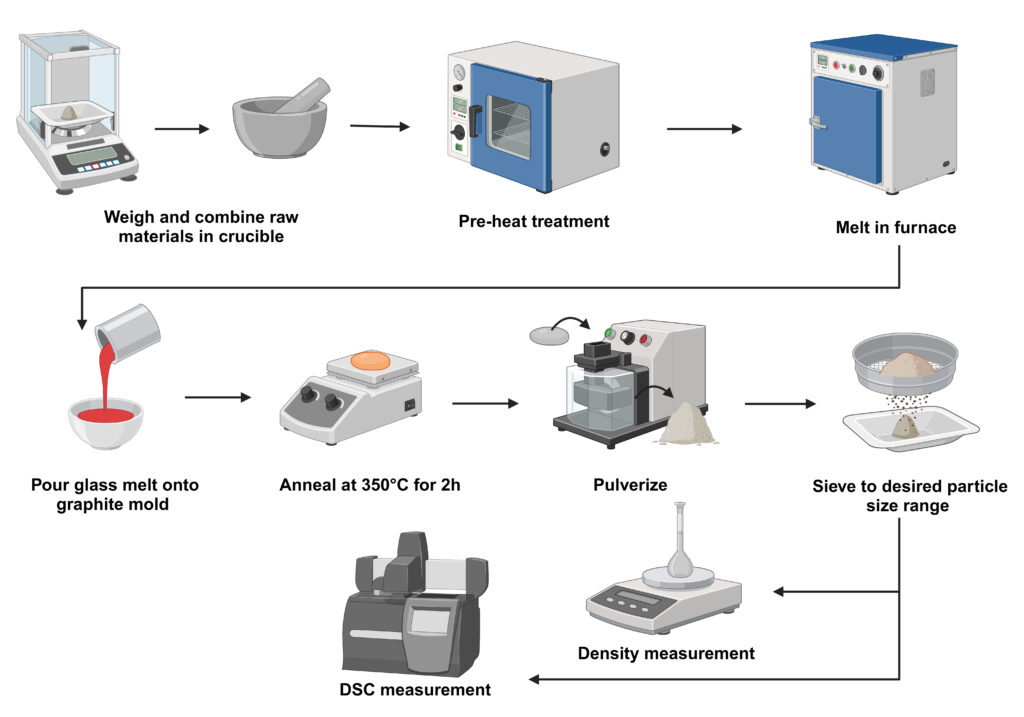
To design bioactive glass compositions with optimal thermal, mechanical, and bioactive properties as coatings on Ti6Al4V metallic implants, we investigated phosphosilicate bioactive glasses based on the 6P55 composition. SiO2 was substituted with B2O3 to improve adhesion to the metallic implants and physical properties. This substitution significantly altered the glass structure and is hypothesized to improve adhesion. Computational and experimental methods revealed that boron substitution introduced BO3 and BO4 units, disrupted the Si-O network, and formed non-bridging oxygens (NBOs), resulting in a decrease in density and glass transition temperature (Tg). These changes were attributed to boron’s dual role as a network former and modifier, influencing coordination environments and connectivity. Thermal and structural analyses showed that optimal boron levels improved thermal expansion and network flexibility, which are critical for coating applications. By integrating molecular dynamics simulations and experimental techniques, this study provides valuable insights into tailoring glass compositions for enhanced performance on metallic substrates.
Significance:
The significance of this research lies in its contribution to designing bioactive glass compositions with tailored thermal, mechanical, and bioactive properties for use as coatings on Ti6Al4V metallic implants. By substituting SiO₂ with B₂O₃ in phosphosilicate bioactive glasses, the study demonstrated how boron incorporation can enhance glass structure and physical properties to improve adhesion and compatibility with metallic implants.
Keywords:
bioactive glass; biomaterials; computational analysis; density functional theory; glass-ceramics; molecular dynamic simulation